Rapid Opioid Cutoff Is Risky Too, Feds Warn

Amid an epidemic of addiction and overdose deaths tied to Oxycontin, Vicodin and other opioid painkillers, federal agencies are urging doctors to evaluate the risks and benefits to patients on high doses of the medicines before abruptly tapering them off. Toby Talbot/AP Photo
Connecting state and local government leaders
Two federal agencies caution doctors not to taper high-dose pain patients too quickly.
This article was originally published by Stateline, an initiative of The Pew Charitable Trusts.
To stem the opioid epidemic, U.S. doctors cut prescriptions of medications such as OxyContin, Vicodin and Percocet by at least a quarter over the last five years. But the reduction in prescriptions came at a cost to some pain patients.
The U.S. Centers for Disease Control and Prevention and the Food and Drug Administration last month warned prescribers that abruptly cutting off high-dose patients or tapering their doses too rapidly could cause withdrawal and even suicide.
The new recommendations likely will prompt states to consider adjusting their opioid prescribing laws, said Karmen Hanson, a public health expert at the National Conference of State Legislatures. But since the CDC and FDA clarifications came near the end of legislative sessions for most states, no actions are anticipated this year, she said.
More than half of states enacted laws restricting painkiller prescriptions after earlier federal guidance urged such limits, but the agencies say that advice may have been misinterpreted or taken too far.
The FDA last month said it had received reports of patients taken off their pain medicines who were suffering “serious withdrawal symptoms, uncontrolled pain, psychological distress and suicide.”
Likewise, the CDC—which in 2016 recommended prescribing limits for opioid painkillers—said it had received reports that some prescribers were too rigidly adhering to its dosage guidelines and had misapplied the limits to patients already on high doses of opioids.
In its 2016 guidelines, the CDC urged doctors to use opioid painkillers sparingly, and instead opt for non-opioid medications and non-pharmacological solutions to pain, such as physical therapy.
The agency recommended a one-week limit on the number of pills prescribed for first-time users suffering from acute pain and urged doctors to avoid increasing longer-term prescriptions for chronic pain to more than the equivalent of 90 mg of morphine a day.
Last month, the agency said: “This statement does not address or suggest discontinuation of opioids already prescribed at higher dosages, yet it has been used to justify abruptly stopping opioid prescriptions or coverage.”
The recommended limit of 90 mg equivalents is roughly equal to two tablets of 30 mg OxyContin or nine tablets of 10 mg Vicodin, a medication level that the agency says should be reserved for cancer patients, patients at the end of life, and others with intractable pain.
The CDC’s clarification came in response to a March letter from a group of more than 300 physicians known as Health Professionals for Patients in Pain who reported that the suggested daily dosage limits had been widely misapplied by doctors, insurers and pharmacists as well as by entire health care systems.
The group, which includes three past U.S. drug czars, said “patients have endured not only unnecessary suffering, but some have turned to suicide or illicit substance use,” because of misapplied CDC guidelines. But they conceded that “experiences for these patients are documented predominantly in anecdotal form.”
Dr. Patrice Harris, president-elect of the American Medical Association, said the CDC’s warning about the guidelines “underscores that patients with acute or chronic pain can benefit from taking prescription opioid analgesics at doses that may be greater than the guidelines.”
The misapplication of the CDC guidelines has been so broad, Harris said, “that it will be hard to undo the damage.”
When the CDC guidelines were published in 2016, the American Medical Association and pain organizations backed by drugmakers complained that the recommendations could make it difficult for chronic pain sufferers to get the pills they need.
Andrew Kolodny, who co-directs opioid research at Brandeis University and heads Physicians for Responsible Opioid Prescribing, said the reports of pain patients suffering because of misapplication of the CDC’s guidelines are exaggerated.
The number of doctors who are inappropriately tapering pain patients is likely very small and should not be blamed on the CDC, he said.
Kolodny said he sympathized with chronic pain patients. “Their emotions are real. But they’re being effectively manipulated to controversialize the CDC guidelines.” He said the CDC’s April statement on high-dose opioid prescribing simply reiterated what it already said in 2016.
‘Pain Refugees’
Representatives of Health Professionals for Patients in Pain praised both federal agencies for issuing clarifications. But they said more needs to be done to reduce prescribers’ concerns that they will run afoul of state and federal regulations if they treat chronic pain patients with the high doses of opioid painkillers that they say many patients need.
Dr. Sally Satel, a psychiatrist and resident scholar at the conservative American Enterprise Institute, who helped organize the group, said that doses as high as 400 to 800 mg morphine equivalents are not unusual for some pain patients.
Next year, Satel said, she expects her group and others will begin talking to state officials about laws that reflect the CDC guidelines.
In most states, laws on opioid prescribing don’t apply to chronic pain patients, she said, “but they certainly reinforce this climate of intense anxiety over the dangers of prescription opioids, which I think has been wildly overstated. Most people can take these meds without problems.”
Satel and other advocates for opioid use in pain management have argued that under pressure from federal, state and health system policies, many doctors have refused to treat pain patients. This has resulted, they said, in an increasing number of what they call pain refugees who go from doctor to doctor searching for someone who will treat their chronic pain.
Kolodny disagrees that the CDC’s guidelines and state prescribing laws are putting too much pressure on prescribers.
“We have a very real problem in this country. But the CDC guidelines didn’t cause it,” he said. “The problem is that millions of Americans have been put on round-the-clock opioids at very high doses and for reasons that doctors now realize were not appropriate.
“What the FDA needs to tell doctors is that because it is so excruciating to come off of opioids, they need to be very selective about who they put on them,” he said. “They need to talk to patients about how they’re going to land the plane before they take off in it.”
Rapid Decline
In an article last month in the New England Journal of Medicine, the authors of the CDC’s guidelines praised efforts by states, hospitals and medical organizations to develop policies that reduce opioid prescribing, addiction and overdoses.
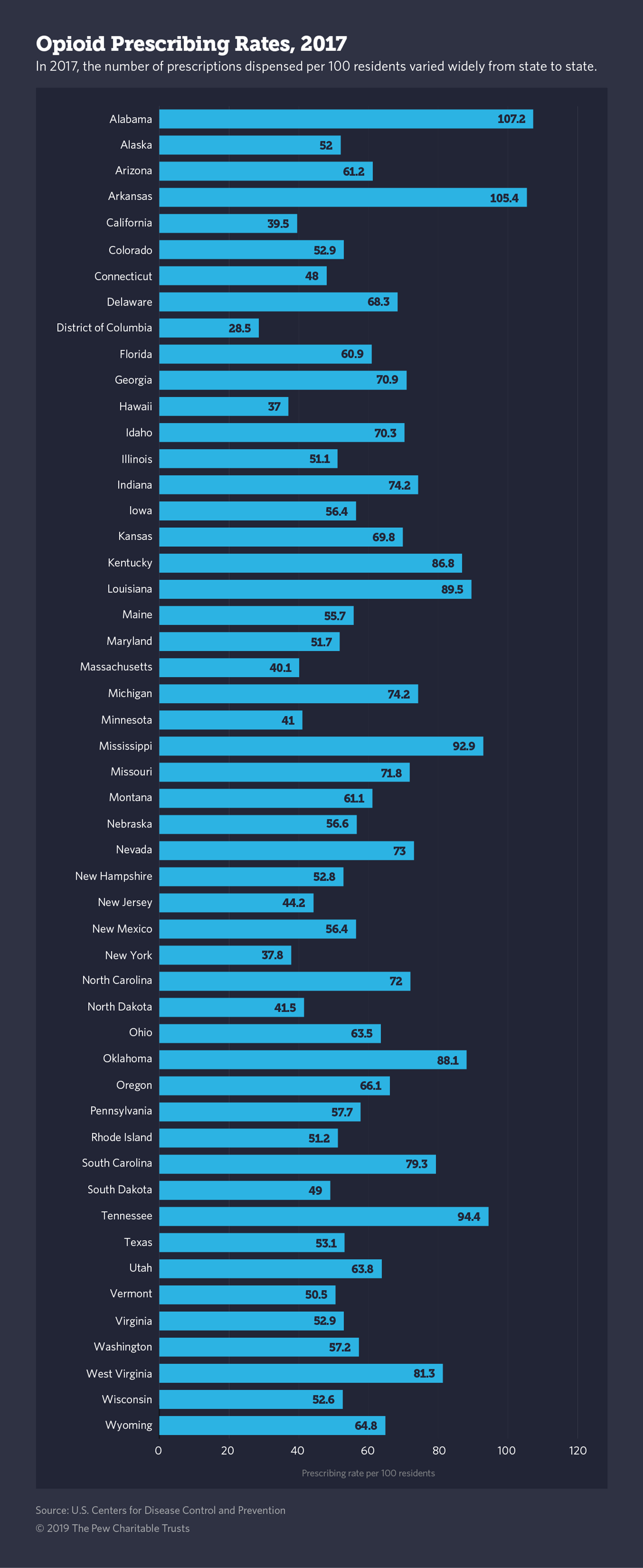
In 2017, the number of opioid prescriptions declined more than 25% from its peak in 2012of 81 per 100 U.S. residents to an average of 59, according to the CDC. During the same period, the rate of high-dose opioid prescribing also declined.
In 2012, 683 people per 100,000 in the United States were prescribed high doses of opioids. By 2016, when the CDC issued its guidelines, the rate of high-dose prescribing already had declined by about 4 per 100,000 each month.
After the guidelines came out, the monthly decline doubled to 8 per 100,000, according to a recent study by University of Michigan researchers.
Another study, by researchers at Harvard, found that first-time opioid prescribing dropped by more than half between 2012 and 2017, largely because many doctors stopped prescribing the painkillers.
Prescribing rates vary widely among states and counties, with some counties having rates seven times the national average, according to the CDC.
State Laws
In 2016, Massachusetts became the first state to enact a law limiting first-time opioid prescriptions to seven days, in line with the CDC’s recommendation.
Since then, 25 states have enacted similar laws restricting opioid prescribing for acute pain.

The strictest limits are in Florida, Kentucky and Tennessee, where initial prescribing is limited to three to four days. Minnesota also limits painkiller prescriptions to three to four days, but only for acute dental or eye pain, according to National Conference of State Legislatures.
Seven other states—New Hampshire, Ohio, Oregon, Vermont, Virginia, Washington state and Wisconsin—have enacted statutes that authorize a state agency to set prescribing limits on opioids.
Arizona and Nevada have laws limiting prescribed doses of opioids to the equivalent of 90 mg a day, as the CDC guidelines recommend. Maine’s limit is 100 mg morphine equivalents.
Rhode Island limits prescriptions to 50 mg morphine equivalents, as does Tennessee, except for prescriptions of three days or less, which can be prescribed at 60 mg equivalents.
Arizona, California, Florida, New Mexico, Ohio, Rhode Island, Tennessee, Vermont, Virginia and Washington state have enacted laws requiring doctors to prescribe or offer to prescribe the overdose rescue drug naloxone to patients who are taking high doses of opioid painkillers. Coprescribing naloxone with high doses of opioid pain medications also was among the CDC’s 2016 guidelines.

NEXT STORY: Tennessee Speaker of the House Announces Resignation


